- Identification
- Unlike its co-occurring congeners Pogonomyrmex maricopa has either no or very short spines on the propodeum.
- Biology
- The venom released by the Maricopa Harvester Ant when stinging is one of the most toxic substances known. Stings by this ant cause a systemic reaction in vertebrates. Such specificity suggests this venom is a compound that has evolved to serve as a defense against vertebrate predators, such as horned lizards, that might target Pogonomyrmex ants as a food resource.
- additional biology notes...
- Distribution
- Range
- United States and Mexico. U.S. - western Texas, New Mexico, southern Colorado, southern Utah, Arizona, southeastern Nevada, southeastern California. Mexico - Sinoloa, Sonora, northern Chihuahua, northern Baja California.
- Navajo Reservation Records
- Samples being processed.
- Additional Notes
- Pogonomyrmex are abundant and often conspicuous ants in arid low-latitude habitats of the Americas. Cole (1968) offered this well written general introduction to the genus:
The genus Pogonomyrmex is the preeminent group of harvesting ants in North America, where it nearly blankets the arid regions of Mexico and the western United States. As is true of other harvesting ants, the workers of this genus collect seeds for food, "harvesting" the plants in their nesting areas by snipping off the seeds with their mandibles. Husked within the nest and stored in subterranean or mound chambers, these highly nutritious seeds become the paramount food for the society, sustaining the ants even through the winter. Seeds are not their sole food, however, because the ants are scavengers as well as harvesters. Although their victims are chiefly arthropods, the workers may transport a varied array of dead booty to their nests.
The nests themselves are constructed in the soil, generally in areas fully exposed to the sun. Some are beneath stones, whereas others are surmounted by soil craters or by small to huge mounds with or without coverings of gravel. In addition, the workers of some species alter the area peripheral to the nests by clearing away the plants, felling them, bit by bit, with their powerful mandibles. Such mounds their surrounding denuded area, characteristic of the western harvester, are familiar sights to travelers in the arid West.
Although the workers of some species of Pogonomyrmex are extremely hostile and will vigorously attack and sting an invader, other species are contrastingly inoffensive and retiring.
- Nesting Biology
- The species commonly occurs in sandy, silt, and loam soils on flat or gently sloping areas. They infrequently occupy rocky areas or steep inclines. Within the range of its suitable nesting substrate, it has a variable nest mound. There may be multiple large sand craters around a number of closely spaced openings, a single sand or pebble crater, or large sand domes that may be covered by pebbles. The common dome-shaped mounds formed by this species are typically surrounded by large bare areas.
- Claustral Nest Founding
- Queens of Pogonomyrmex maricopa are provisioned in their natal colony with enough reserves to begin a new colony without needing to forage (Johnson 2002). An average of 7 minims is produced by a queen in a newly founded nest.
- Foraging
- Foraging activity during the summer is limited to daylight hours and begins after sunrise. Activity peaks within a few hours of start-up, with a gradual decline in the number of foragers as the temperature increases through to midday. Foraging slows to low levels but does not stop through the heat of the day, slightly increases through the afternoon, and stops altogether in the early evening. The ability of Pogonomyrmex maricopa to forage through the heat of the day is reflected in workers of this species possessing a high LD50 (a measure of the temperature that is lethal to workers).
- Pogonomyrmex maricopa individuals may leave the nest in any direction, unlike other Pogonomyrmex species that use trunk trails for directing their foraging, i.e., Pogonomyrmex barbatus. Pogonomyrmex maricopa are not strong recruiters. Workers from neighboring colonies will fight fiercely when they meet at the periphery of the foraging area that surrounds their nests.
- The maximum foraging distance from the colony entrance is 25 m. Workers rely strongly on visual cues to find their way back to their nest. Seeds comprise the majority of items collected by foragers, with insects also being important.
- See Hansen 1978, Hölldobler 1974, Hölldobler 1976b for more information.
- Mating
- Mating flights are initiated by the arrival of rains during the rainy season. The day after the first substantial rain, workers and alates will congregate around the nest entrance in the morning and fly away in the late morning (at least around Portal, AZ). Males congregate in large numbers on a tree or bush, 2 to 4 m in height, with one such plant or groups of plants being located up to a kilometer or more from other similar lek sites. The males release a secretion from their mandibular gland that serves as a pheromone for attracting females. Virgin females fly into these mating swarms, where they are quickly set upon by numerous eager-to-mate males. Up to four males will simultaneously compete for copulations with a single female. New males replace departing but successfully mated males in quick succession, leading to polygyny being the norm for Pogonomyrmex maricopa queens.
- After mating, females fly off and attempt to locate a suitable place for colonization. They will land on the ground in a favorable place and quickly pull off their wings, climb up on the vegetation, find a suitable resting place and remain there for many hours. This behavior presumably saves the females from having to begin digging a nest in the hot ground during the heat of the day.
- See Holldobler, 1976a for more information.
- Etymology
- The name Maricopa was originally applied to a southwestern Tribe of the Yuman people. Wheeler was likely picking up on the common usage of this name in this region.
- Moody and Foster
- The biology of this species was detailed in a study by Moody and Foster (1979) that was published in a National Park Service Transactions bulletin focused on the Guadalupe Mountains. All of the text that follows in this section is from this publication.
- Pogonomyrmex maricopa constructs a dome-shaped mound surrounded by a bare area of 4 to 10ft (120 x 0300cm) in diameter. The mound is constructed from excavated soil supplemented with twigs, pebbles, and miscellaneous particles returned by foraging workers. Completed mounds stand 12 to 18in. (30 to 45 cm) high and range from 24 to 30 in. (60 to 75 cm) in base diameter.
- Very young colonies lack the characteristic mound and bare area. They surround their nest entrance with twigs and debris. As excavation and expansion proceed, the mound slowly becomes more typical in appearance. Mounds are evident in second year colonies. The development of the bare area corresponds with the construction of the mound.
- The nest entrance is located usually 2 to 3 in. (5 to 7.5 cm) above the base of the east-facing mound surface. Occasionally, a nest has two separate entrances. If so, they are close together and open into the same gallery. The entrance is roughly circular and large enough to accommodate two or three ants simultaneously. The position of the entrance facilitates warming in the early morning and shading in the late afternoon. This allows workers to begin foraging early in the morning and to resume foraging earlier in the afternoon.
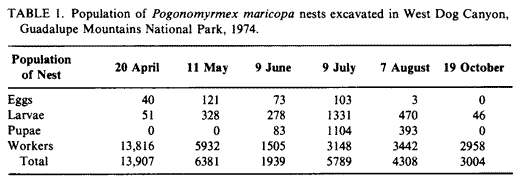
- Although colonies with approximately equal mound and base area size were selected for excavation, populations varied greatly (Table I). There seems to be no correlation between mound size and ant numbers in colonies 2 years old or older. Growing colonies sometimes abandon nests in favor of nearby, larger vacant nests. The age structure of each nest population excavated was recorded (Table I). Reproductive activity had begun prior to the first excavation as indicated by the presence of eggs and larvae in the nest. Pupae were encountered first on 9 July; no teneral adults were present. It is interesting to note that egg production peaked during June. The number of both larvae and pupae were greatest in July.
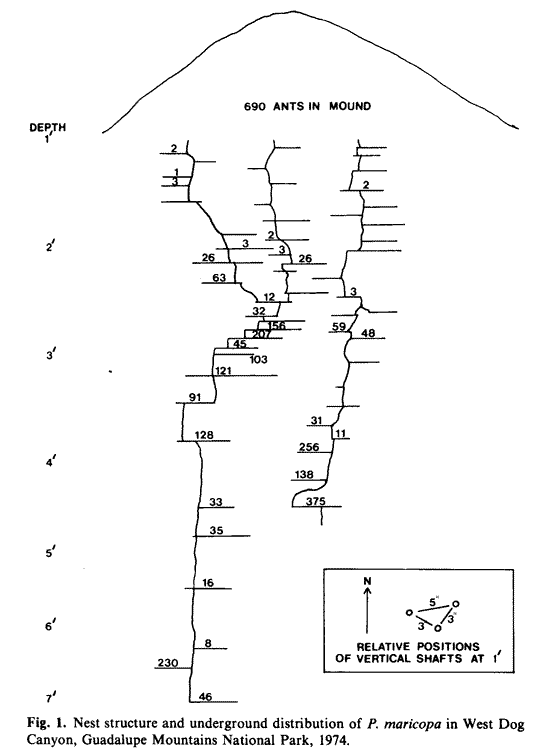
- Winged adults were present in July and appeared above ground in large numbers at the time of the first rains. Reproduction declined sharply during the latter part of the summer as reflected by egg, larval, and pupal populations encountered during August. By October, the only immatures present were a few larvae scattered throughout the nest. The nest excavated on 19 October 1974 (Fig. I) was typical both in structure and population distribution.
- Abandoned trash and storage galleries in the honeycomb area occasionally were inhabited by larvae of Collops sp. (Coleoptera: Malachiidae). Collops larvae are predaceous and probably feed on scavengers associated with these galleries. Another insect found in the honeycomb area was the larva of Gonasida elata (LeConte) (Coleoptera: Tenebrionidae), which apparently spends its entire larval life burrowing through the mound.
- Foraging activity is affected strongly by surface temperature. The nest entrance usually is uncovered and the first workers emerge when the surface temperature at the nest opening reaches 21 to 23°C. Activity increases until the surface temperature reaches 40 to 45°C and decreases sharply at temperatures above 46°C. Activity is reduced greatly at temperatures above 57°C.
- On clear days foraging usually was terminated by 11a.m. and began again when temperatures dropped to the 48 to 52°C range. Afternoon activity ceased when temperatures dropped to the upper 20s. This bimodal pattern of daily activity did not occur on cloudy days when the soil surface was exposed only to brief periods of direct sunlight.
- It was noted on several occasions that rain caused the cessation of foraging activity. During light rain, workers stopped leaving the mound; foragers returning with booty were unaffected. During heavy rains, foragers returned directly to the nest. Termination of light rain brought a return of normal foraging activity. During the first drought-breaking rains of July, large numbers of workers massed around their nest entrances, where they imbibed water for several minutes at a time. Such behavior did not occur during subsequent rains in July and August.
- Foraging patterns suggest that populations in the study area forage from their mounds in all directions. Ants generally cross the bare area by fairly well-defined routes, which lead in four or five directions. Beyond the bare area, movement becomes less directional. Foraging distances sometimes extend as far as 41ft (12.3 m) from the nest entrance. However, most forage excursions did not extend more than 16 to 20 ft (4.8 to 6 m) from the entrance. Foraging is terminated when the first acceptable booty is encountered. Returning ants usually follow a more direct route.
- Booty returned by foraging ants during the early afternoons of 9 July, 3 August, and 18 October were collected and analyzed. On 9 July a few days after the rainy season had begun, insect parts comprised 35%of the material returned, whereas seeds accounted for 54%. Twigs and pebbles accounted for only 8%. On l August, seeds and insect parts accounted for only 32%of the booty; twigs and pebbles comprised 68%. Mound repair appeared to be the prime function of workers visible during the study period. On 18 October, the colony was engaged in gathering provisions. Sixty-two percent of the material returned was seeds, whereas insect parts comprised only 2%. Twigs and pebbles accounted for 35%.
- Observations made over the entire season suggest that seeds are gathered whenever they are available, and that most plants within the forage range are utilized. Insect parts comprise a substantial part of the forage material only after the initial summer rains, which trigger a flush of insect emergence. Twigs, dirt, and pebbles are gathered most frequently during late afternoon. These materials are applied directly to mound-building or repair.

as reported by Cole (1968)
- Literature
- Hansen, S. R. 1978. Resource utilization and coexistence of three species of Pogonomyrmex ants in an Upper Sonoran grassland community. Oecologia (Berlin). 35:109-117.
- Hölldobler, B. 1974. Home range orientation and territoriality in harvesting ants. Proceedings of the National Academy of Sciences of the United States of America. 71:3274-3277.
- Hölldobler, B. 1976a. The behavioral ecology of mating in harvester ants (Hymenoptera: Formicidae: Pogonomyrmex). Behavioral Ecology and Sociobiology. 1:405-423.
- Hölldobler, B. 1976b. Recruitment behavior, home range orientation and territoriality in harvester ants, Pogonomyrmex. Behavioral Ecology and Sociobiology. 1:3-44.
- Johnson, R. A. 2002. Semi-claustral colony founding in the seed-harvester ant Pogonomyrmex californicus: a comparative analysis of colony founding strategies. Oecologia. 132:60-67.
- Moody, J. V. and D. E. Forster. 1979. Notes on the bionomics and nest structure of Pogonomyrmex maricopa (Hymenoptera: Formicidae). National Pks. Serv. Trans. Proc. Ser. No. 4: 115-121.
- Wheeler, W. M. 1914. New and little known harvesting ants of the genus Pogonomyrmex. Psyche (Cambridge). 21:149-157.
- A note about these publications. The literature cited here is not meant to be an exhaustive list of papers published about this species.
Page authored by David Lubertazzi and Gary Alpert